Introduction
In this tutorial we describe a full Bayesian framework for species tree estimation. The statistical methodology described in this tutorial is known by the acronym *BEAST (pronounced “star beast”) (Heled & Drummond, 2010).
You will need the following software at your disposal:
- BEAST - this package contains the BEAST program, BEAUti, TreeAnnotator and other utility programs. This tutorial is written for BEAST v2.4.x, which has support for multiple partitions. It is available for download from http://beast2.org (Bouckaert et al., 2014).
- Tracer - this program is used to explore the output of BEAST (and other Bayesian MCMC programs). It summarizes graphically and quantitively the distributions of continuous parameters and provides diagnostic information for the particular MCMC chain. At the time of writing, the current version is v1.6. It is available for download from http://tree.bio.ed.ac.uk/software/tracer.
- FigTree - this is an application for displaying and printing molecular phylogenies, in particular those obtained using BEAST. At the time of writing, the current version is v1.4.2. It is available for download from http://tree.bio.ed.ac.uk/software/figtree.
*BEAST analysis
This tutorial will guide you through the analysis of three loci sampled from 26 individuals representing nine species of pocket gophers. This is a subset of previously published data (Belfiore et al., 2008). The objective of this tutorial is to estimate the species tree that is most probable given the multi-individual multi-locus sequence data. The species tree has nine taxa, whereas each gene tree has 26 taxa. *BEAST 2 will co-estimate three gene trees embedded in a shared species tree (see (Heled & Drummond, 2010) for details).
The first step in the analysis will be to convert a NEXUS file with a DATA or CHARACTERS block into a BEAST XML input file. This is done using the program BEAUti (Bayesian Evolutionary Analysis Utility). This is a user-friendly program for setting the evolutionary model and options for the MCMC analysis. The second step will be to actually run BEAST using the XML input file that contains the data, model and MCMC chain settings. The final step will be to explore the output of BEAST in order to diagnose problems and to summarize the results.
BEAUti
Start up BEAUti to set up the configuration file for the analysis.
Set up BEAUti for *BEAST
BEAUti uses templates to populate the BEAST XML configuration file with the necessary elements. This template also allows BEAUti to set up the graphical user interface accordingly, having the right tabs and settings.
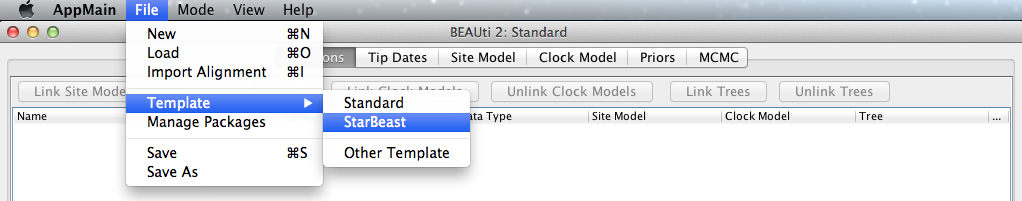
*BEAST uses a non-standard template to generate the analysis XML, which means that to use BEAUti for *BEAST, the first thing to do is to change the template. Choose the File/Template/StarBeast item (Figure 1). Keep in mind that when changing a template, BEAUti deletes all previously imported data and starts with a clean template. So, if you already loaded some data, a warning message will pop up indicating that this data will be lost if you switch templates.
Loading the NEXUS file
*BEAST is a multi-individual, multi-locus method. The data for each locus is stored as one alignment in its own NEXUS file. Taxa names in each alignment have to be unique, but duplicates across alignments are allowed.
To load a NEXUS format alignment, simply select the Import Alignment option from the File menu. Select three files called 26.nex, 29.nex, 47.nex by holding shift key.
You can find the files in the examples/nexus directory in the directory where BEAST was installed.
Alternatively, click the links above to download the data. After the data is opened in your web browser each time, right click with the mouse and separately save them as 26.nex, 29.nex, 47.nex.
Each file contains an alignment of sequences from an independent locus. The 26.nex looks like this (content has been truncated):
#NEXUS
[TBO26oLong]
BEGIN DATA;
DIMENSIONS NTAX =26 NCHAR=614;
FORMAT DATATYPE = DNA GAP = - MISSING = ?;
MATRIX
Orthogeomys_heterodus ATTCTAGGCAAAAAGAGCAATGC ...
Thomomys_bottae_awahnee_a ????????????????????ATGCTG ...
Thomomys_bottae_awahnee_b ????????????????????ATGCTG ...
Thomomys_bottae_xerophilus ????????????????????ATGCTG ...
Thomomys_bottae_cactophilus ????????????????AGCAATGCT ...
... ...
;
END;
Once loaded, the three partitions are displayed in the main panel. You can double click any alignment (partition) to show its detail. Figure 2 shows the way BEAUti looks after loading the appropriate alignments.
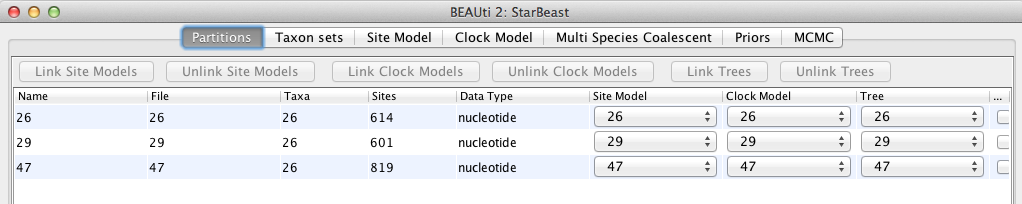
For multi-locus analyses, BEAST can link or unlink substitutions models across the loci by clicking buttons on the top of Partitions panel. The default of *BEAST is unlinking all models: substitution model, clock model, and tree models. For this tutorial we shall leave all models unlinked.
Note that you should only unlink the tree model across data partitions that are actually genetically unlinked. For example, in most organisms all the mitochondrial genes are effectively linked due to a lack of recombination and they should be set up to use the same tree model in a *BEAST analysis. It could also be that for this analysis a linked model would perform better.
Guess or import trait(s) from a mapping file
Each taxon in a *BEAST analysis is associated with a species. Typically, the species name is already embedded inside the taxon. The species name should be easy to extract; place it either at the beginning or the end, separated by a “special” character which does not appear in names. For example, aria_334259, coast_343436 (using an underscore) or 10x017b.wrussia, 2x305b.eastis (using a dot).
We need to tell BEAUti somehow which lineages in the alignments go with taxa in the species tree. Select the Taxon Set panel, and a list of taxa from the alignments is shown together with a default guess by BEAUti. In this case, the guess is not very good, so we want to change this. You can either change each of the entries in the table manually, have a mapping stored in a file or have BEAUti guess the taxon names.
If the mapping is to be read from a file one needs to specify a proper trait file. The first row is always specified as the keyword traits in the first column followed by the keyword species in the second column, separated by tab. The rest of the rows map each individual taxon name to a species name; the taxon name in the first column and species name in the second column separated by tab. For example:
traits species
taxon1 speciesA
taxon2 speciesA
taxon3 speciesB
... ...
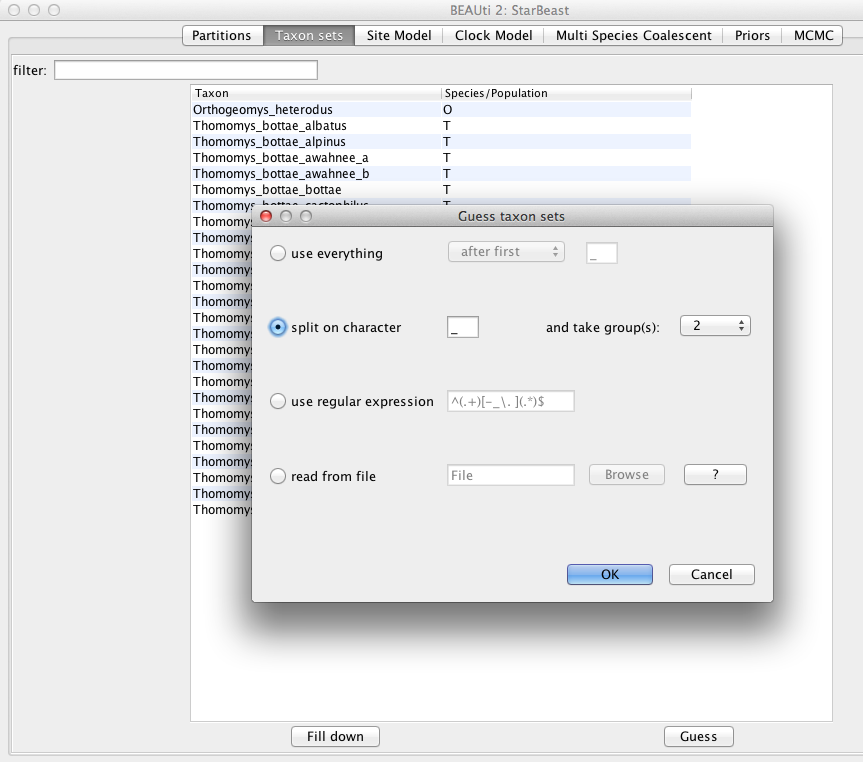
Alternatively, pressing the Guess button in BEAUti, a dialog is shown where you can choose from several ways to try to detect the species from the name of the taxon. In our case, splitting the name on the underscore character (_) and selecting the second group will give us the mapping that we need (Figure 3).
Setting up the substitution model
The next thing to do is to click on the Site Model tab at the top of the main window. This will reveal the evolutionary model settings for BEAST. Exactly which options appear depends on whether the data are nucleotides, amino acids, binary data, or general data. The settings that appear after loading the dataset are always the default values.
Thus, we have to pick a substitution model that will work better on real data than the default JC69.
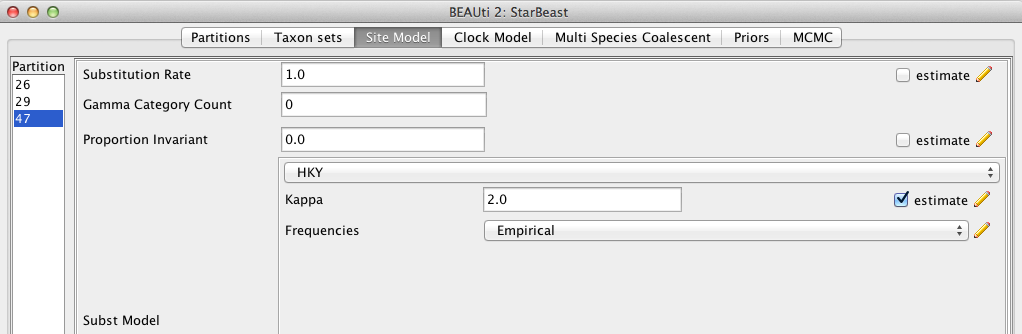
For this analysis, we select each partition of the data on the left side of the panel and set up the same substitution model for all three partitions: HKY for Substitution Model and Empirical for the Frequencies (Figure 4).
Setting up the clock model
Then, click on the Clock Model tab at the top of the main window. In this analysis, we use the Strict Clock molecular clock model.
Your model options should now look as displayed in Figure 5.
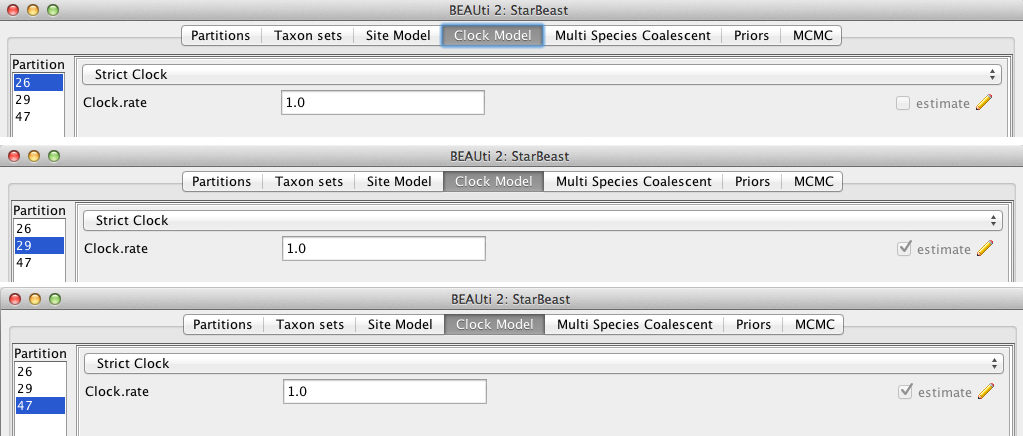
The estimate check box is unchecked for the first clock model and checked for the rest clock models, because we wish to estimate the substitution rate of each subsequent locus relative to the first locus whose rate is fixed to 1.0.
Multi Species Coalescent
The Multi Species Coalescent panel allows settings of the multi-species coalescent model to be specified. Users can only configure the species tree prior, not the gene tree priors, which are automatically specified by the multispecies coalescent.
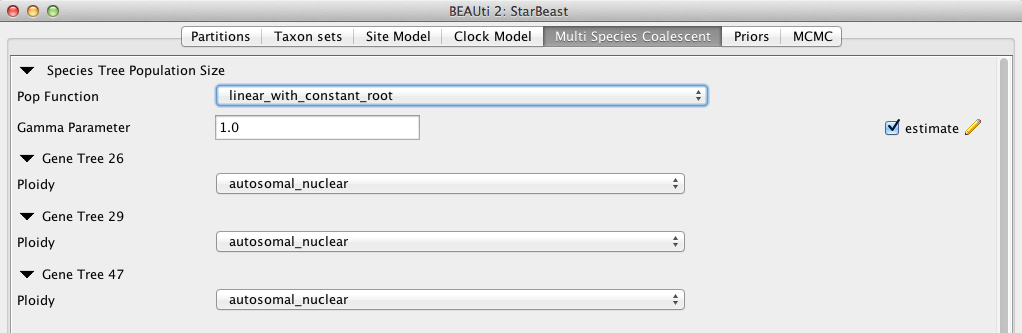
Currently, three population size models for species tree (under the entry Pop Function) are available: Continuous-linear and constant root, Continuous-linear, and Constant. In this analysis, since we don’t have much information for the population change at the root, we use linear and constant root model.
In *BEAST, the population sizes are assigned gamma priors, and users cannot change the type to a different distribution. But the scale parameter of the gamma prior (called Population Mean) can be assigned a hyperprior (tick estimate, Figure 6).
The Ploidy item determines the type of sequence (mitochondrial, nuclear, X, Y). This matters since different modes of inheritance give rise to different effective population sizes, for example in the case of mitochondrial DNA we would only deal with the half of the available population, since this type of DNA is only passed on by the mother. In the case of this analysis the data is autosomal nuclear, which is the default option (Figure 6).
Priors
The Priors panel allows priors for each parameter in the model to be specified.
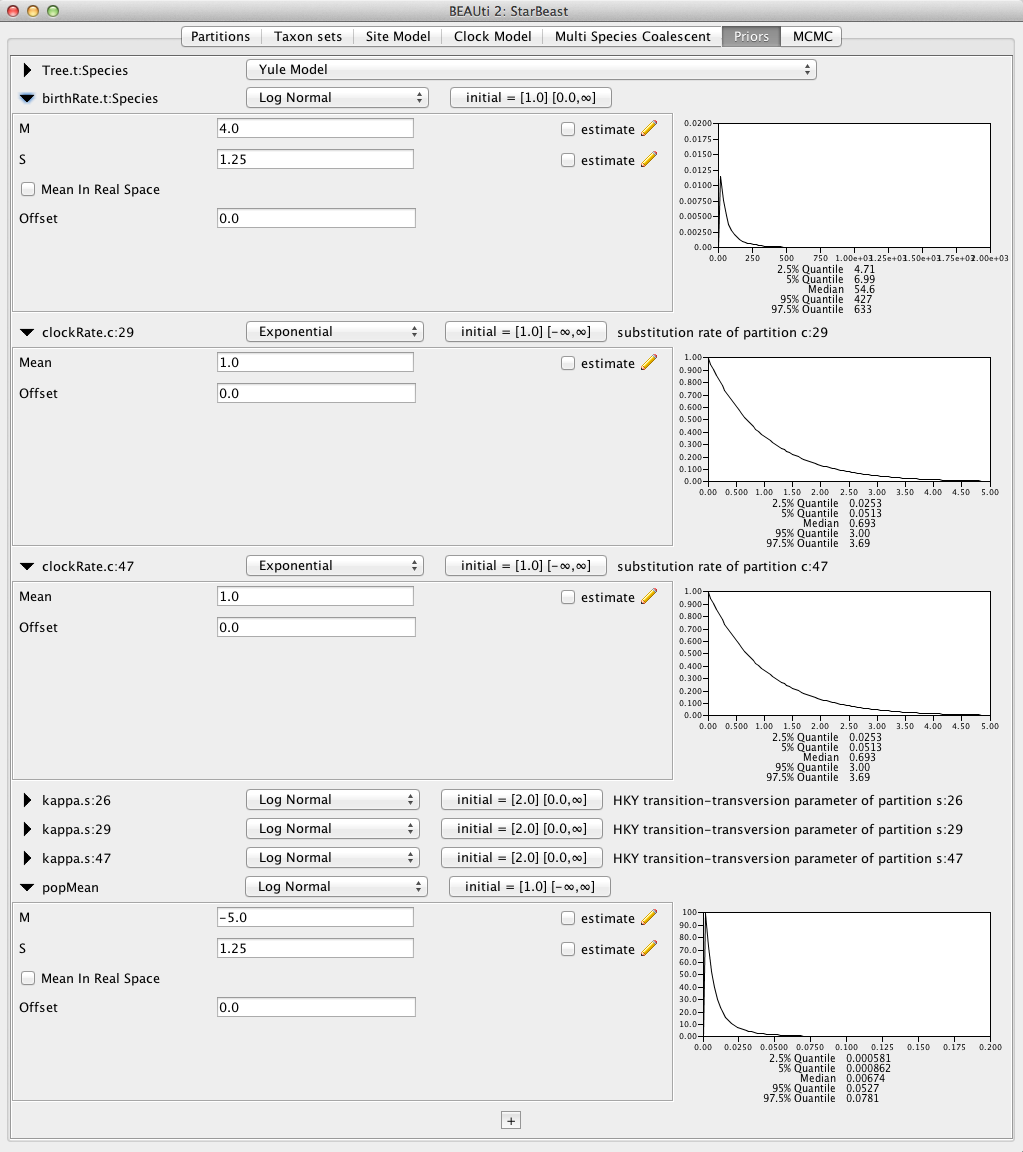
Several models are available as species tree priors, however, only the Yule or the birth-death model are suitable for *BEAST analyses. Leave the tree prior setting at the default Yule Model. This prior specifies the distribution of the speciation times.
The default priors that BEAST sets for other parameters would allow the analysis to work. However, some of these are inappropriate for this analysis. Therefore change the priors as follows:
- birthRate.t:Species: set the prior to
Log Normalwith M of 4.0 and S of 1.25; - popMean (scale parameter of the gamma priors for population sizes): set the prior to
Log Normalwith M of -5.0 and S of 1.2; - clockRate.c:29: set the prior to
Exponentialwith mean 1.0; - clockRate.c:45: set the prior to
Exponentialwith mean 1.0.
The resulting configuration is shown in Figure 7.
Operators
The Operators panel (normally hidden) is used to configure technical settings that affect the efficiency of the MCMC program. In MCMC the operators define which values will be proposed for the next step in the chain given the current state. You can take a look at them if you go to View/Show Operators panel, but do not edit the settings.
Setting the MCMC options
The next tab, MCMC, provides more general settings to control the length of the MCMC and the file names.
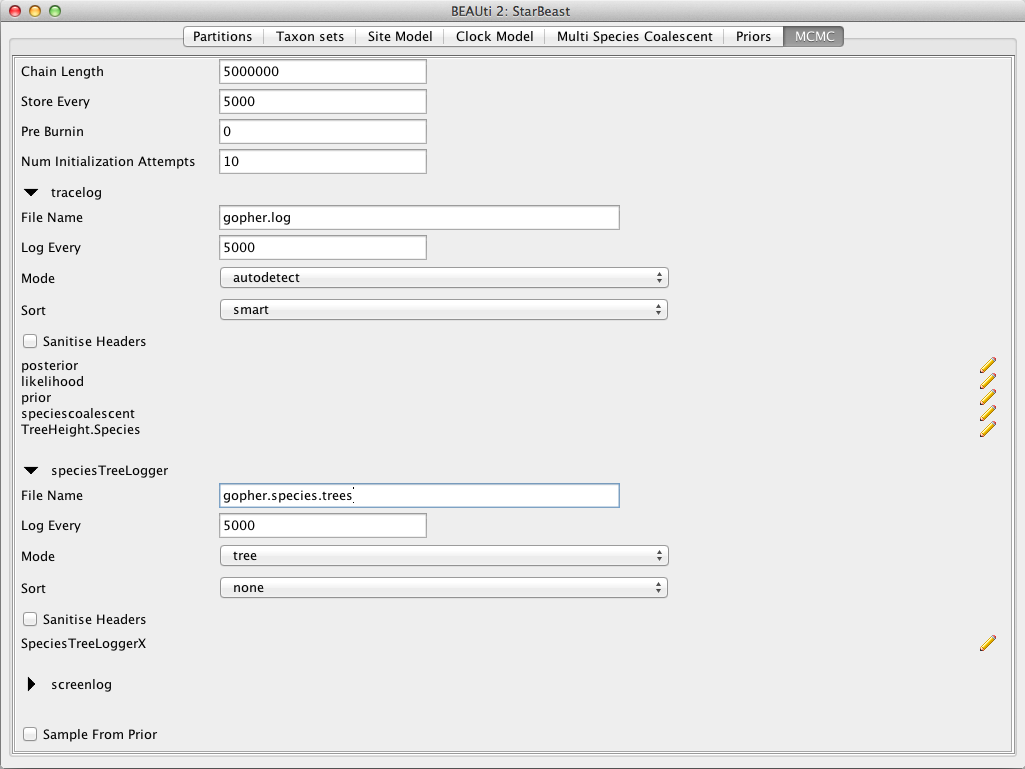
First, we need to set the Chain Length. This is the number of steps the MCMC will make in the chain before finishing. The appropriate length of the chain depends on the size of the dataset, the complexity of the model and the accuracy of the answer required. The default value of 10,000,000 is entirely arbitrary and should be adjusted according to the size of your dataset. For this dataset let’s set the chain length to 5,000,000 as this will run reasonably quickly on most modern computers (a few minutes).
The next options specify how often should the parameter values in the Markov chain be displayed on the screen and recorded in the log file. The screen output is simply for monitoring the program’s progress and can thus be set to any value (although if set too small, i.e. if displaying on screen too often, the sheer quantity of information being displayed on the screen will actually slow the program down). For the log file, the value should be set relative to the total length of the chain. Sampling too often will result in very large files with little extra benefit in terms of the precision of the analysis. Sample too infrequently and the log file will not contain much information about the distributions of the parameters. You probably want to aim to store no more than 10,000 samples so the log frequency should be set to no less than 5,000,000 / 10,000 = 500.
For this exercise we will set the the tracelog to 5,000 and the screenlog to 10,000.
Set the file name of the tracelog to gopher.log, that of the speciesTreeLogger to gopher.species.trees, and that of the treelog.t:26, treelog.t:29, treelog.t:47 to gopher.gene$(tree).trees.
The resulting configuration is shown in Figure 8.
If you are using Windows then we suggest you add the suffix .txt to both of these files (i.e. to specify output file names to gopher.log.txt and gopher.species.trees.txt) so that Windows recognizes these as text files.
Generating the BEAST XML file
We are now ready to create the BEAST XML file. To do this, either select the File/Save or File/Save As option from the File menu. Save the file with an appropriate name (we usually end the filename with .xml, i.e. gopher.xml). We are now ready to run the file through BEAST.
BEAST
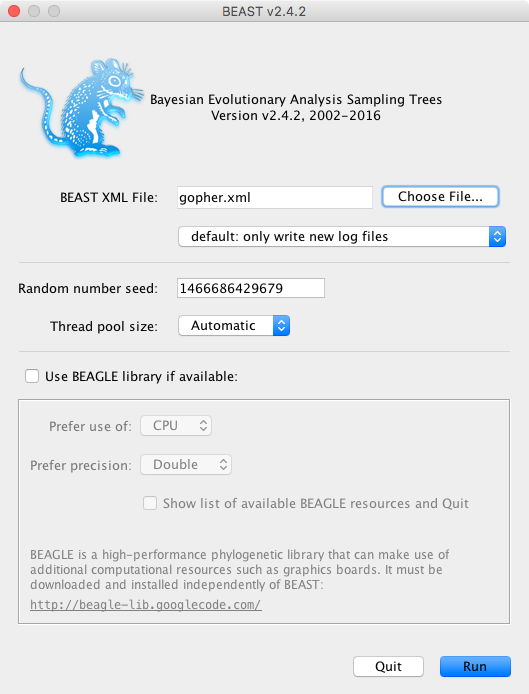
Now run BEAST. Provide your newly created XML file as input by clicking Choose File …, and then click Run (Figure 9).
BEAST will then run until the specified chain length is reached, and until it has finished reporting information on the screen. The actual result files are saved to the disk in the same location as your input file. The output to the screen will look something like this:
BEAST v2.4.2, 2002-2016
Bayesian Evolutionary Analysis Sampling Trees
Designed and developed by
Remco Bouckaert, Alexei J. Drummond, Andrew Rambaut & Marc A. Suchard
Department of Computer Science
University of Auckland
remco@cs.auckland.ac.nz
alexei@cs.auckland.ac.nz
Institute of Evolutionary Biology
University of Edinburgh
a.rambaut@ed.ac.uk
David Geffen School of Medicine
University of California, Los Angeles
msuchard@ucla.edu
Downloads, Help & Resources:
http://beast2.org/
Source code distributed under the GNU Lesser General Public License:
http://github.com/CompEvol/beast2
BEAST developers:
Alex Alekseyenko, Trevor Bedford, Erik Bloomquist, Joseph Heled,
Sebastian Hoehna, Denise Kuehnert, Philippe Lemey, Wai Lok Sibon Li,
Gerton Lunter, Sidney Markowitz, Vladimir Minin, Michael Defoin Platel,
Oliver Pybus, Chieh-Hsi Wu, Walter Xie
Thanks to:
Roald Forsberg\includeimage, Beth Shapiro and Korbinian Strimmer
... ...
4980000 -3827.2093 279.2 -4293.2486 22.1146 24s/Msamples
4990000 -3842.9689 276.3 -4295.6213 17.6877 24s/Msamples
5000000 -3798.1962 278.2 -4280.7865 22.0904 24s/Msamples
Operator Tuning #accept #reject Pr(m) Pr(acc|m)
NodeReheight(Reheight.t:Species) - 205317 486572 0.1386 0.2967
ScaleOperator(popSizeBottomScaler.t:Species) 0.1902 10224 26338 0.0074 0.2796
ScaleOperator(popMeanScale.t:Species) 0.4890 5795 16242 0.0044 0.2630
UpDownOperator(updown.all.Species) 0.5029 38885 108717 0.0295 0.2634
ScaleOperator(YuleBirthRateScaler.t:Species) 0.2416 7155 15125 0.0044 0.3211
ScaleOperator(treeScaler.t:26) 0.7580 8510 42962 0.0102 0.1653
ScaleOperator(treeRootScaler.t:26) 0.4424 11487 39392 0.0102 0.2258
Uniform(UniformOperator.t:26) - 287517 225590 0.1024 0.5603
SubtreeSlide(SubtreeSlide.t:26) 0.5560 418 256419 0.0512 0.0016 Try decreasing size to about 0.278
Exchange(narrow.t:26) - 115438 140923 0.0512 0.4503
Exchange(wide.t:26) - 1363 49988 0.0102 0.0265
WilsonBalding(WilsonBalding.t:26) - 1882 49890 0.0102 0.0364
ScaleOperator(StrictClockRateScaler.c:29) 0.4744 13355 37595 0.0102 0.2621
ScaleOperator(treeScaler.t:29) 0.7567 8137 42785 0.0102 0.1598
ScaleOperator(treeRootScaler.t:29) 0.4101 12191 38887 0.0102 0.2387
Uniform(UniformOperator.t:29) - 296262 216280 0.1024 0.5780
SubtreeSlide(SubtreeSlide.t:29) 0.5724 339 255443 0.0512 0.0013 Try decreasing size to about 0.286
Exchange(narrow.t:29) - 117912 138109 0.0512 0.4606
Exchange(wide.t:29) - 2229 49303 0.0102 0.0433
WilsonBalding(WilsonBalding.t:29) - 2459 49001 0.0102 0.0478
UpDownOperator(updown.29) 0.7793 10314 40694 0.0102 0.2022
ScaleOperator(StrictClockRateScaler.c:47) 0.5490 12974 38097 0.0102 0.2540
ScaleOperator(treeScaler.t:47) 0.7363 8746 42523 0.0102 0.1706
ScaleOperator(treeRootScaler.t:47) 0.4742 7774 43670 0.0102 0.1511
Uniform(UniformOperator.t:47) - 276942 234361 0.1024 0.5416
SubtreeSlide(SubtreeSlide.t:47) 0.5044 307 255119 0.0512 0.0012 Try decreasing size to about 0.252
Exchange(narrow.t:47) - 91846 164747 0.0512 0.3579
Exchange(wide.t:47) - 731 50281 0.0102 0.0143
WilsonBalding(WilsonBalding.t:47) - 937 50377 0.0102 0.0183
UpDownOperator(updown.47) 0.7397 10853 40537 0.0102 0.2112
UpDownOperator(strictClockUpDownOperator.c:47) 0.7278 9987 41373 0.0102 0.1945
UpDownOperator(strictClockUpDownOperator.c:29) 0.7761 10040 40724 0.0102 0.1978
ScaleOperator(KappaScaler.s:26) 0.3401 469 1197 0.0003 0.2815
ScaleOperator(KappaScaler.s:29) 0.2955 436 1231 0.0003 0.2615
ScaleOperator(KappaScaler.s:47) 0.3199 379 1298 0.0003 0.2260
ScaleOperator(popSizeTopScaler.t:Species) 0.1910 23665 54936 0.0157 0.3011
Tuning: The value of the operator's tuning parameter, or '-' if the operator can't be optimized.
#accept: The total number of times a proposal by this operator has been accepted.
#reject: The total number of times a proposal by this operator has been rejected.
Pr(m): The probability this operator is chosen in a step of the MCMC (i.e. the normalized weight).
Pr(acc|m): The acceptance probability (#accept as a fraction of the total proposals for this operator).
Total calculation time: 135.584 seconds
End likelihood: -3798.196207797654
Analyzing the results
Tracer
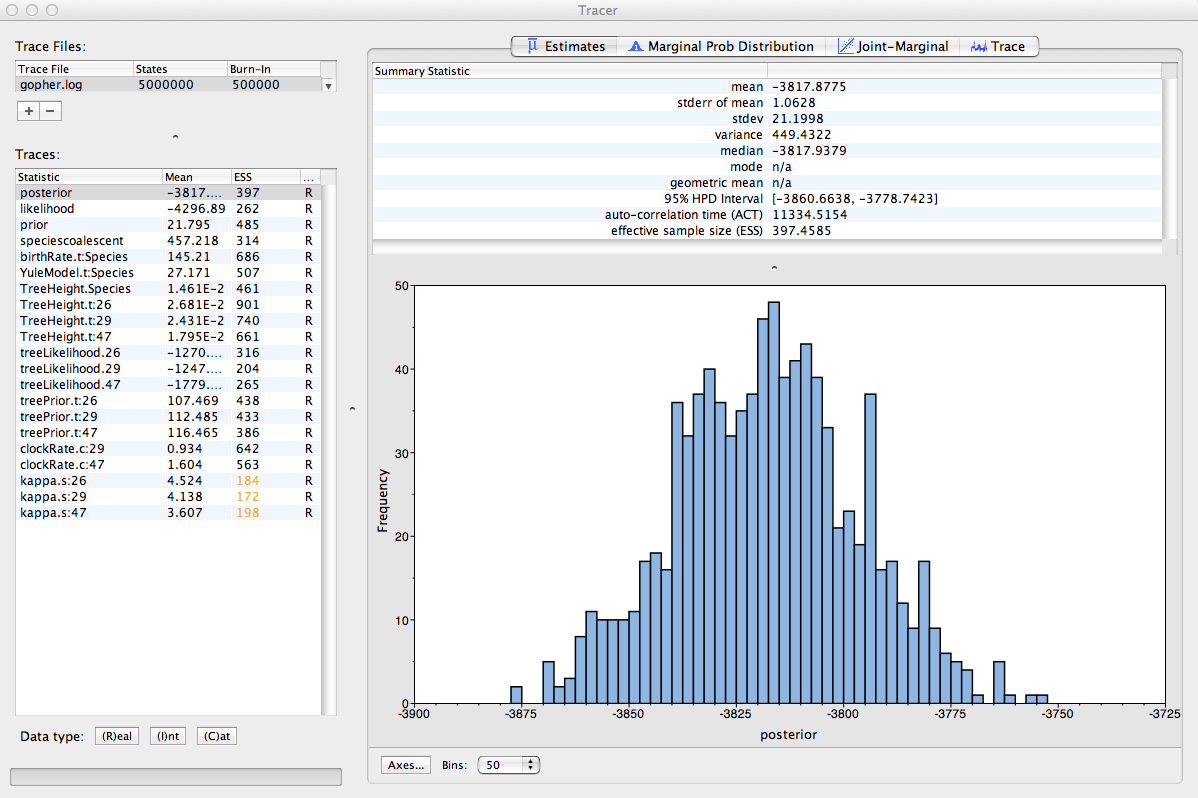
Run the program called Tracer to analyze the output of BEAST. When the main window has opened, choose Import Trace File… from the File menu and select the file that BEAST has created called gopher.log.
You should now see a window looking like the snapshot in Figure 10.
Remember that MCMC is a stochastic algorithm so the actual numbers will not be exactly the same.
On the left-hand side is a list of the different quantities that BEAST has logged. There are traces for the posterior (this is the log of the product of the likelihood and the prior probabilities), and the continuous parameters. Selecting one trace from this list on the left-hand side brings up analyses for this trace on the right-hand side. When a file is first opened in Tracer, the posterior trace is selected and various statistics of this trace are shown under the Estimates tab (top window on the right-hand side).
Tracer will plot a (marginal posterior) distribution for the selected parameter and also give you statistics such as the mean and median. The 95% HPD lower or upper stands for highest posterior density interval and represents the most compact interval on the selected parameter that contains 95% of the posterior probability. It can be thought of as a Bayesian analog to a confidence interval.
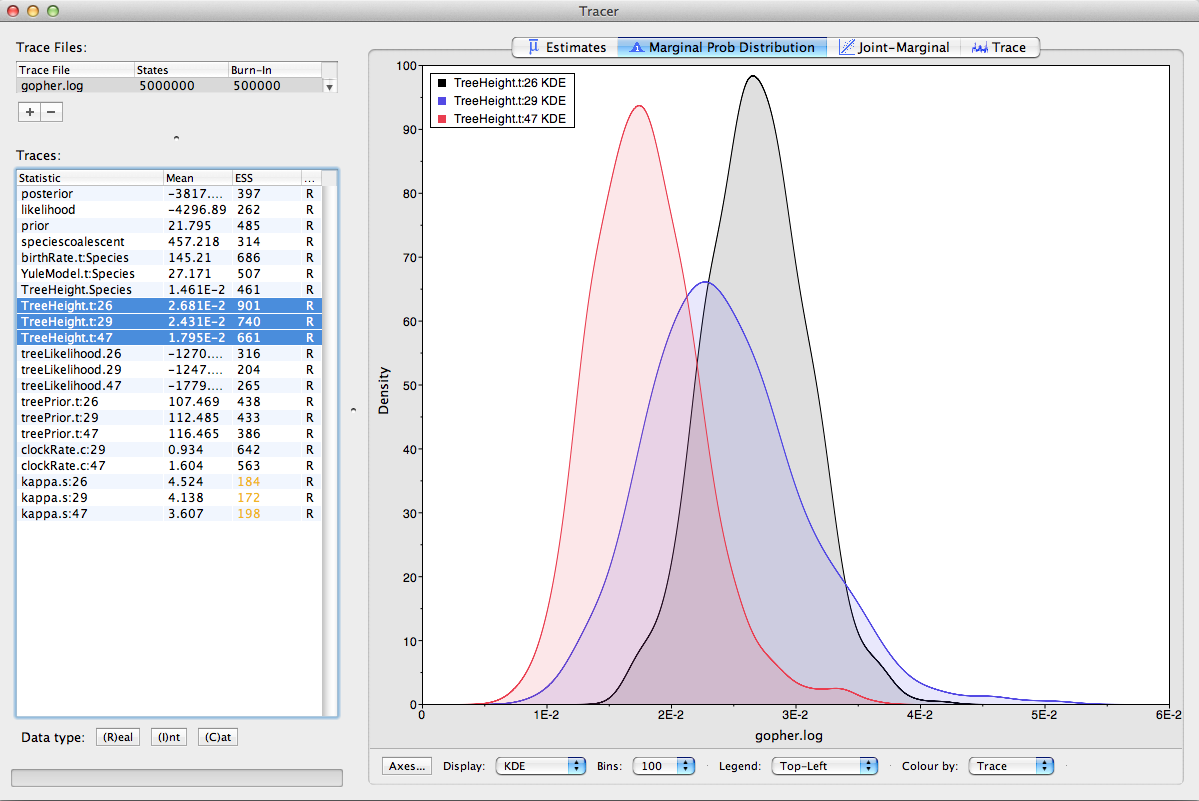
Select the three TreeHeight traces (hold `shift key while selecting). This will display the
age of the root for the three gene trees. If you switch the tab at the top of the window on the right-hand side to Marginal Prob Distribution then you will get a plot of the marginal posterior densities of each of these date estimates overlayed (Figure 11).
TreeAnnotator
BEAST also produces a sample of plausible trees. These can be summarized using the program TreeAnnotator. This program will take the set of trees and identify a single tree that best represents the posterior distribution. It will then annotate this selected tree topology with the mean ages of all the nodes as well as the 95% HPD interval of divergence times for each clade in the selected tree. It will also calculate the posterior clade probability for each node. Run the TreeAnnotator program and set it up as shown in Figure 12.
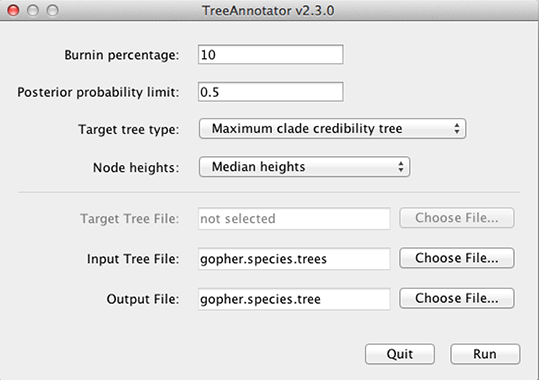
The Burnin percentage is the number of trees to remove from the start of the sample. Unlike in Tracer where burnin is specified as the number of steps, in TreeAnnotator you need to specify the percentage of trees discarded. For this run, we set the burnin to 10 (i.e. we remove first 10% of the trees).
The Posterior probability limit option specifies a limit such that if a node is found at less than this frequency in the sample of trees (i.e., has a posterior probability less than this limit), it will not be annotated. Setting this to zero will annotate all nodes. For this analysis, set it to 0.5. This means that only the nodes seen in the majority of trees will be annotated.
For Target tree type you can either choose a specific tree from a file or ask TreeAnnotator to find a tree in your sample. The default option, Maximum clade credibility tree, finds the tree with the highest product of the posterior probability of all its nodes.
Choose Median heights for Node Heights. This sets the heights (ages) of each node in the tree to the median height across the entire sample of trees for that clade.
For the input file, select the trees file that BEAST created (in this tutorial this was the gopher.species.trees) and select a file for the output (here we called it gopher.species.tre).
Now press Run and wait for the program to finish.
Viewing the species tree
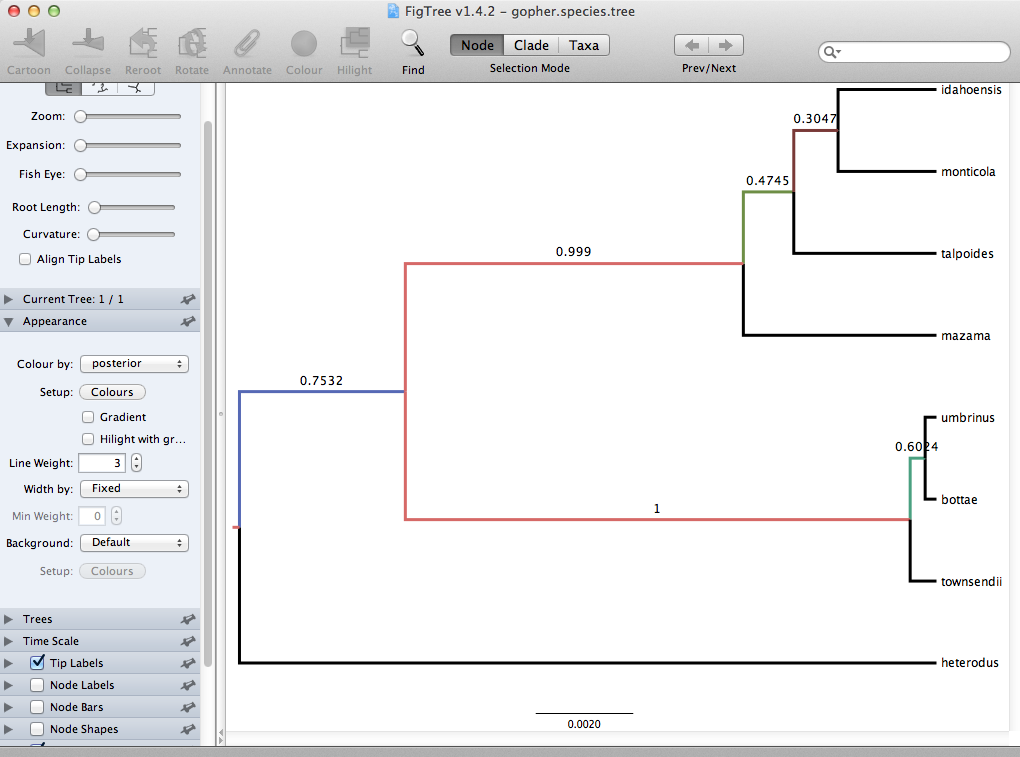
Finally, we can look at the tree in another program called FigTree. Run this program, and open the gopher.species.tre file by using the Open command in the File menu. The tree should appear.
You can now try selecting some of the options in the control panel on the left. Tick the Node Bars and try to get node age error bars. Also turn on Branch Labels and select posterior to get it to display the posterior probability for each node. Under Appearance you can also tell FigTree to colour the branches by the `posterior.
You should end up with a tree similar to one displayed in Figure 13.
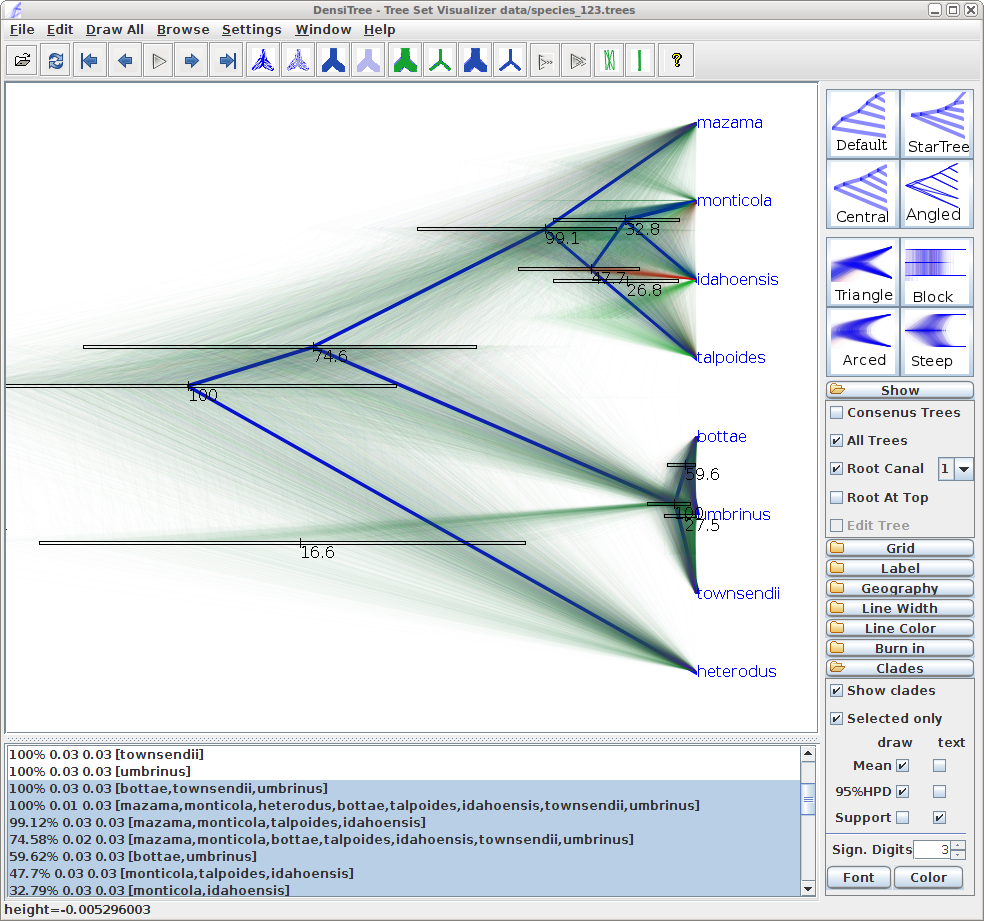
Alternatively, you can load the species tree set gopher.species.trees into DensiTree and visualize it with following settings.
-
Perhaps, the intensity of the individual trees is not high enough, so you might want to increase it by clicking the icon Increase Intensity of Trees in the button bar at the top. The text for each button shows when you hover over it.
-
Set Burn in to 500 trees. If before you had a collapsed trees, after this setting the tree should not be collapsed anymore.
-
Show a root-canal tree to better guide the eye over the set of trees.
-
In Clades menu, select Show clades and display their mean and 95% HPD graphically, and posterior support using text. By default you may not be able to select the Show clades option. If that is the case, first click on the icon Central in the top right corner of the window.
You can see that there is information for too many clades being displayed. As most clades have very little support, they are not of interest and we would like to stop visualizing the information for those. Tick Selected only, then open the clade toolbar (menu Window/View clade toolbar), and select only highly supported clades (hold down theshiftkey to select several clades at a time). Also, select the clade consisting of heterodus, bottae, umbinus and townsendii, to show that heterodus is an outgroup of the tree, but there is some support ( 17%) that it is not. -
Drag the clades monticola and idahoensis up in the displayed tree so that the 95% HPD bar does not overlap with the one for mazama, monticola, idahoensis and talpoidis. Increase font size of the label for better readability (i.e. change the Label options).
You should now see an image similar to Figure 14.
DensiTree can be used to show the population sizes of the each of the trees from gopher.species.trees as branch widths.
Under Line Width, select BY_METADATA_PATTERN option, for both upper and lower spinners and use .*dmv=.(^,*).* for the upper (start of the branch) pattern and .*dmv=.^,*,(^\}*).* for the lower (end of the branch) pattern.
This gives us visualisation as seen in Figure 15.

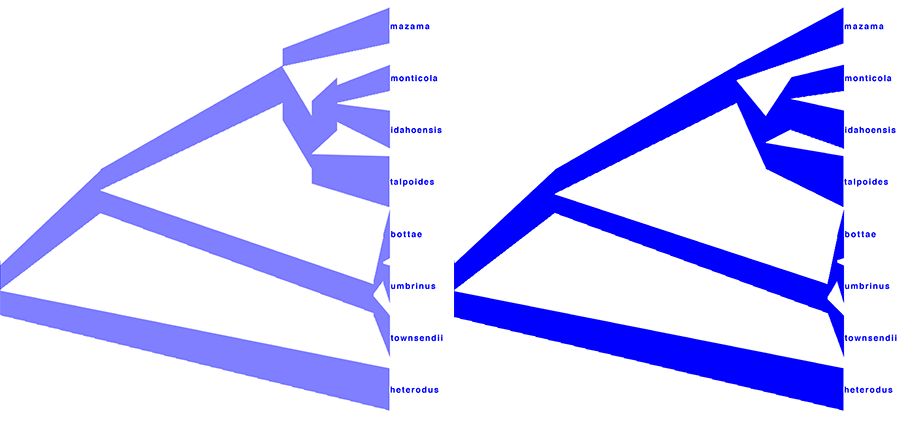
Similarly, DensiTree can be used to show the population sizes of the summary tree gopher.species.tre from TreeAnnotator.
Load this file to DensiTree and explore the options in the spinners under Line Width. Notice, that dmt stands for demographic times, dmv for demographic values. These are the population parameters that are estimated for each node in the tree. With the continous-linear population size model, the first value dmv1 is the population size at the branch start and the second dmv2 is the population size at the branch end; where start and end are relative to going backwards in time, that is start is the more recent time, end the more ancient.
Choose dmv1 for the upper (start/bottom of a branch) and dmv2 for the lower spinner (end/top of a branch).
The resulting tree should look like the one in the left picture in Figure 16.
You can see that at the nodes, the population size estimates do not quite match.
This is true for the areas of the tree with little posterior support for the clades (see Figure 14 to check which clades have little support). In the right picture in Figure 16 the top is matched up with the bottom of the branch above (more ancestral), using the Make fit to bottom option for the lower spinner. This looks a bit prettier, but may not be quite accurate.
Alternatively to TreeAnnotator, a consensus tree can be generated by biopy. An example is shown in Figure 17 where 1-norm option has been applied to generate the left picture, and 2-norm to obtain the right picture.

Comparing your results to the prior
Using BEAUti, set up the same analysis but under the MCMC options, select the Sample From Prior option. Re-run the analysis with BEAST. This will allow you to visualize the full prior distribution in the absence of your sequence data. Summarize the trees from the full prior distribution and compare the summary to the posterior summary tree.
You can also compare the log files from both analyses by loading the log file from the actual analysis and the log file from the prior sampling run into Tracer. Then select both of the files on the left side using the shift key. Now you can select different statistics from the traces and compare their marginal probability distributions. Note that Tracer will not allow you to compare any of the likelihoods as they are always NaN (not a number) in the case of prior sampling.
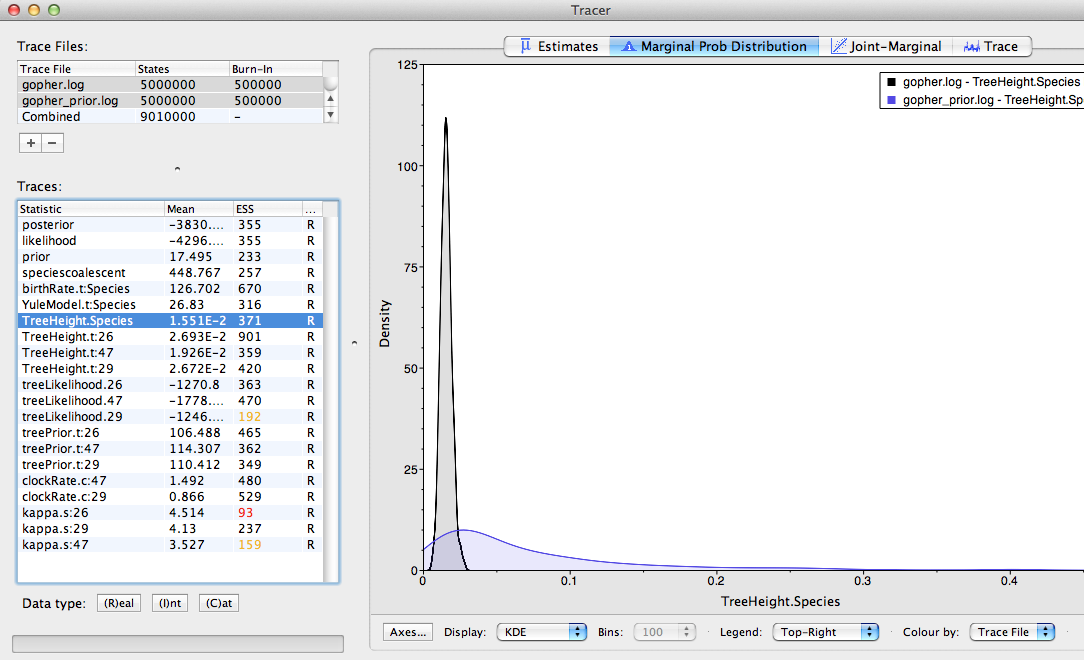
Figure 18 shows an example of comparing the traces of the species tree height. From the plot we can conclude that the sequences contain quite a bit of information about this parameter, as the posterior moved away from the prior and is much more focused on a specific value.
The content of this tutorial is based on the *BEAST in BEAST 2.0 tutorial by Joseph Heled, Remco Bouckaert, Alexei J. Drummond and Walter Xie.
Relevant References
- Heled, J., & Drummond, A. J. (2010). Bayesian inference of species trees from multilocus data. Mol Biol Evol, 27(3), 570–580. https://doi.org/10.1093/molbev/msp274
- Bouckaert, R. R., Heled, J., Kühnert, D., Vaughan, T., Wu, C.-H., Xie, D., Suchard, M. A., Rambaut, A., & Drummond, A. J. (2014). BEAST 2: a software platform for Bayesian evolutionary analysis. PLoS Comput Biol, 10(4), e1003537. https://doi.org/10.1371/journal.pcbi.1003537
- Belfiore, N. M., Liu, L., & Moritz, C. (2008). Multilocus phylogenetics of a rapid radiation in the genus Thomomys (Rodentia: Geomyidae). Systematic Biology, 57(2), 294.
Citation
If you found Taming the BEAST helpful in designing your research, please cite the following paper:
Joëlle Barido-Sottani, Veronika Bošková, Louis du Plessis, Denise Kühnert, Carsten Magnus, Venelin Mitov, Nicola F. Müller, Jūlija Pečerska, David A. Rasmussen, Chi Zhang, Alexei J. Drummond, Tracy A. Heath, Oliver G. Pybus, Timothy G. Vaughan, Tanja Stadler (2018). Taming the BEAST – A community teaching material resource for BEAST 2. Systematic Biology, 67(1), 170–-174. doi: 10.1093/sysbio/syx060